Sample preparation by borate fusion and X-Ray Fluorescence (XRF) spectrometry has been provent many times to be a robust analytical method. This paper examines an optimum analytical method that was developed using an automated fusion instrument, Claisse® M4TM fluxer (Figure 1), for sample preparation, and a Wavelength Dispersive X-Ray Fluorescence (WDXRF) spectrometer for determination of all the relevant elements used in the global cement industry.
This approach was utilized to prepare process materials, end products, and a wide range of raw materials. Some of the raw materials covered by this analytical application are cement, aluminate cement, cements with additions, blended cement, limestone, sand, gypsum, bauxite, kiln feed, and sand clinker amongst others. In addition, the sample preparation was optimized for the TheOx® fluxer by performing an accurate substantiation of the method's performance.

Figure 1. Claisse® M4™ Fluxer
Since powders are affected by particle size and mineralogy effects, it is very difficult to utilize a single XRF calibration curve to examine different materials through the pressed powder preparation method. However, when fused in a borate glass, such particle size and mineralogy effects can be removed. Analysis of raw materials can be performed with a single XRF calibration curve covering different range of concentrations for all relevant elements in the cement industry.
This article explores all the XRF analytical conditions for the calibration of the whole range of raw materials using the borate fusion methodology and also the various Reference Materials (RMs) employed for this analytical application. The results of the XRF raw materials calibration are presented with respect to accuracy, precision, and limit of detection.
Experimental Framework
Apparatus and Instrumental Conditions
For the study, a Bruker-AXS S4 Explorer sequential WDXRF spectrometer with a rhodium end-window X-ray tube of 1000W was utilized to produce data. Parameters such as the spectrometer analytical conditions, background position, background measurements, pulse-height, peak- line, and counting time were optimized by the wavelength step-scanning of the selected standard disks.
The analytical lines for specific elements were introduced to the analysis method since the reference values for these elements exist in the RMs. Subsequently, a 28mm collimator mask was used to perform all the measurements under vacuum.
Calibration Preparation
The calibration of the WDXRF raw materials application was carried out by utilizing a wide range of RMs from the following origins:
- Domtar® Inc. Research Center: Canadian Certified Reference Materials
- Bureau of Analysed Samples Ltd. (BAS): British Chemical Standard Certified Reference Materials
- European Committee for Iron and Steel Standardization
- China National Analysis Center for Iron and Steel: NCS DC Reference Material
- Japan Cement Association (JCA): Reference Materials for X-ray Fluorescence Analysis
- Geological Institute for Chemical Minerals: GBW Reference Material
- European Coal and Steel Community (ECSC): Euro-Standard
- Instituto de Pesquisas Tecnológicas (IPT) : Reference Material
- Institut de Recherches de la Sidérurgie (IRSID) : Échantillon-Type
- South Africa Bureau of Standards: SARM Certified Reference Material
- Slovak Institute of Metrology (SMU): Slovak Reference Material
- National Institute of Standards & Technology (NIST): Standard Reference Material®
Table 1. shows the certified element concentration ranges in the ignited base as well as in the original sample base.
Table 1. RM element concentration as an oxide equivalent
| Compound |
Concentration Range of the Certified Reference Materials |
| Original Sample Base(%) |
LOI Free Base(%) |
| SiO2 |
0,02 - 99,78 |
0,03 - 99,86 |
| Al2O3 |
0,004 - 85,07 |
0,005 - 85,32 |
| Fe2°3 |
0,005 - 85,3 |
0,005 - 91,03 |
| CaO |
0,006 - 70 |
0,006 - 98,58 |
| MgO |
0,001 - 21,25 |
0,001 - 39,66 |
| SO3 |
0,02 - 46,3 |
0,02 - 58,54 |
| Na2O |
0,001 - 4,81 |
0,001 - 4,84 |
| K2O |
0,001 - 4,99 |
0,001 - 5,02 |
| TiO2 |
0,004 - 3,76 |
0,004 - 3,77 |
| P2O5 |
0,003 - 8,42 |
0,003 - 8,62 |
| Mn2O3 |
0,0001 - 4,93 |
0,0002 - 5,05 |
| SrO |
0,001 - 0,638 |
0,001 - 0,649 |
| Cr2O3 |
0,0002 - 0,474 |
0,0004 - 0,486 |
| ZnO |
0,0001 - 0,107 |
0,0001 - 0,109 |
| ZrO2 |
0,005 - 0,14 |
0,005 - 0,2 |
| V2O5 |
0,0006 - 0,72 |
0,0007 - 0,75 |
| BaO |
0,0012 - 0,66 |
0,0012 - 0,66 |
Two individual sets of the different standard glass disks were created on the Claisse TheOx® and the M4TM Classie fluxers®. The first set was utilized for calibration. After the calibration was over, the two sets of glass disks were examined as unknown. The results thus obtained were utilized to assess the accuracy and precision of the method.
Results and Discussion
The Fusion Methodology
During the analytical process of a global fusion method, it was ascertained that sample ignition is necessary. This is a major step and enables fusion of the cements with additions as well as raw materials, which are not easy to fuse in the non-ignited state using conventional fusion techniques.
For preparation with a sample to flux ratio of 1:10, a fusion program of 13 minutes of heating at 1050°C is required to prepare stable glass disks with high silica or high alumina samples. Following this, the cooling process is achieved with forced air in 5 minutes.
In order to show the robustness of this fusion method, over 200 different samples from 20 different types of materials were combined with the global fusion method. Table 2 shows a list of materials comprising only the cements with known additions; the two slag samples that failed included a higher level of copper; and the iron ore samples that failed also included a higher level of copper.
Table 2. List of materials used in this experiment
| # |
Material Type |
Tried |
Success |
# |
Material Type |
Tried |
Success |
| 1) |
Cement |
118 |
118 |
9) |
Bauxite |
4 |
4 |
| 2) |
Cement with AdditionsA |
15 |
15 |
10) |
Sand |
7 |
7 |
| 3) |
Aluminate Cement |
7 |
7 |
11) |
Silica Fume |
3 |
3 |
| 4) |
Clinker |
13 |
13 |
12) |
Fly Ash |
9 |
9 |
| 5) |
Kiln Feed/ Raw Mix |
11 |
11 |
13) |
SlagB |
8 |
6 |
| 6) |
Limestones |
9 |
9 |
14) |
Iron OreC |
19 |
12 |
| 7) |
Gypsum |
7 |
7 |
15) |
Other |
5 |
5 |
| 8) |
Clay |
7 |
7 |
|
Overall |
242 |
233 |
A. Only the cements with known additions are listed here; the cement category probably included some cements with additions
B. The two slag samples that failed contained a higher level of copper
C. The iron ore samples that failed contained a higher level of copper
The global fusion methodology is suitable for preparing stable and uniform lithium borate glass disks with all the materials, except for copper slags, copper-rich iron ores, and iron ores with high magnetite content.
Calibration
Table 3. shows the inter-element corrections as well as their type. It also shows the squared correlation coefficients from the calibration curves of the inspected elements for optimized selection of RMs.
Table 3. Inter-element corrections and squared correlation coefficients for raw materials application
| Element Line |
Inter-element Correction Information |
Squared Correlation Coefficient |
| Al Kα |
Fixed Alphas |
--- |
1,0000 |
| Ba Lα |
Fixed Alphas |
Overlap for Ti |
0,9982 |
| Ca Kα |
Fixed Alphas |
--- |
1,0000 |
| Cr Kα |
Fixed Alphas |
Overlap for V |
0,9997 |
| Fe Kα |
Variable Alphas |
--- |
1,0000 |
| K Kα |
Fixed Alphas |
--- |
0,9999 |
| Mg Kα |
Fixed Alphas |
--- |
0,9999 |
| Mn Kα |
Fixed Alphas |
Overlap for Cr |
0,9998 |
| Na Kα |
Fixed Alphas |
--- |
0,9998 |
| P Kα |
Fixed Alphas |
--- |
1,0000 |
| S Kα |
Variable Alphas |
--- |
1,0000 |
| Si Kα |
Variable Alphas |
--- |
1,0000 |
| Sr Kα |
Fixed Alphas |
--- |
0,9995 |
| Ti Kα |
Fixed Alphas |
--- |
0,9999 |
| V Kα |
Fixed Alphas |
Overlap for Ba & Ti |
1,0000 |
| Zn Kα |
Fixed Alphas |
--- |
0,9986 |
| Zr Kα |
Fixed Alphas |
Overlap for Sr |
0,9988 |
From the calibration of different types of elements, three correlation curves of corrected concentrations against certificate concentrations are of significant interest (Figures 2 to 4).
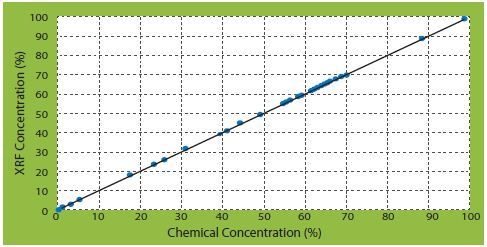
Figure 2. CaO correlation curves of corrected concentrations vs certificate concentrations
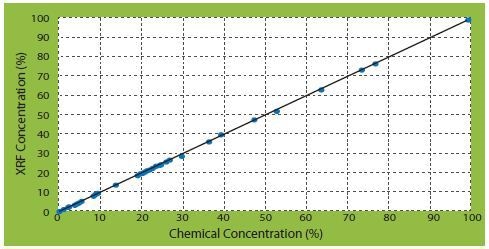
Figure 3. SiO2 correlation curves of corrected concentrations vs certificate concentrations
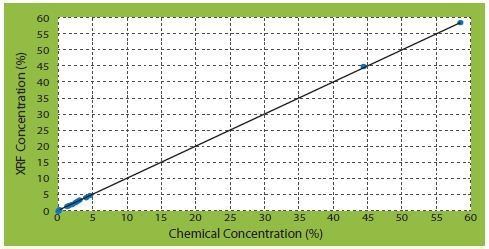
Figure 4. SO3 correlation curves of corrected concentrations vs certificate concentrations
Table 4. shows the results acquired by evaluating accuracy, precision, and sensitivity. By means of the spectrometer software, the results of the sensitivity were obtained using the Lower Limit of Detection (LLD).
Precision was assessed on an absolute concentration base (%) by estimating the maximum difference between the results of the inspected elements for duplicate preparations of all reference materials utilized in the calibration.
The precision evaluation was carried out for the TheOx® and the M4TM fluxers, and the accuracy evaluation was ascertained on an absolute concentration base (%) by measuring the maximum difference of the two results acquired from the duplicates on both the TheOx® and the M4TM fluxer, against the qualified value across all the reference materials utilized in the calibration. The standard deviation determined by the software is also shown in Table 4. The results showed excellent precision and accuracy in spite of the extensive range of elements.
Table 4. Raw materials application results
| Compound |
LLD (ppm) |
Max. Dev.A Between Duplicates M4TM (%) |
Max. Dev.A Between Duplicates TheOx® (%) |
Software Standard Deviation (%) |
Max. Dev.A from Certified Value (%) |
| SiO2 |
40 |
0,10 |
0,15 |
0,16 |
0,59 |
| Al2O3 |
61 |
0,11 |
0,16 |
0,105 |
0,34 |
| Fe2O3 |
51 |
0,12 |
0,11 |
0,089 |
0,36 |
| CaO |
41 |
0,17 |
0,19 |
0,16 |
0,34 |
| MgO |
84 |
0,04 |
0,04 |
0,081 |
0,22 |
| SO3 |
47 |
0,11 |
0,05 |
0,062 |
0,19 |
| Na2O |
85 |
0,02 |
0,02 |
0,018 |
0,07 |
| K20 |
17 |
0,02 |
0,02 |
0,011 |
0,04 |
| TiO2 |
42 |
0,02 |
0,01 |
0,0130 |
0,06 |
| P2O5 |
58 |
0,02 |
0,03 |
0,0074 |
0,03 |
| Mn2O3 |
25 |
0,009 |
0,012 |
0,012 |
0,04 |
| SrO |
14 |
0,01 |
0,006 |
0,0044 |
0,014 |
| Cr2O3 |
34 |
0,006 |
0,005 |
0,0024 |
0,008 |
| ZnO |
13 |
0,003 |
0,002 |
0,0010 |
0,003 |
| ZrO2 |
11 |
0,002 |
0,002 |
0,0032 |
0,007 |
| V2O5 |
15 |
0,003 |
0,001 |
0,0019 |
0,004 |
| BaO |
57 |
0,01 |
0,006 |
0,0060 |
0,014 |
A. Max. Dev. = Maximum Deviation.
Conclusion
This article described the global fusion and XRF analytical methodology for cement industry materials. The sample preparation by fusion methodology helps in fusing raw materials and cements that are typically found in cement factories.
The cement calibration conforms to the accuracy and precision requirements of the International Standard Methods for cement analysis (ISO/DIS 29581-2 and ASTM C114). The study shows that it is possible to achieve an accurate universal calibration, covering the wide range of raw materials utilized in the cement industry.

This information has been sourced, reviewed and adapted from materials provided by Claisse.
For more information on this source, please visit Claisse.