Initially, oil analysis was used for engines as a predictive maintenance tool. To date, it is a method widely used for ensuring the reliability of engine systems. Most of the mobile equipment across the globe (for example, agricultural equipment, mining equipment, trucks, locomotives, cars, buses, and also oil and gas exploration rigs, stationary backup power generators, and pipeline compression stations) are powered by reciprocating internal combustion engines.
Ask the Expert: Engine Oil Analysis
Engine oil must be changed before it loses its capacity to sufficiently perform its intended functions inside an engine. However, frequent or early changing of oil could lead to a huge environmental and cost impact. Oil in rotating internal combustion engines (RICE) gets slowly contaminated, and the contamination rate can vary based on age, load factor, environment, duty cycle, and fuel types. Generally, engine oils contain additive packages composed of alkaline components and detergents for neutralizing acids formed due to the combustion of fuel. The depletion of additives occurs to the point where the combination of the additive and the oil cannot sufficiently protect the engine. In general, it is significant to know the type of contaminant in the oil as it provides symptoms of the engine condition to enable a more directed remedy to take corrective action. For instance, high levels of glycol contamination suggest a leak in the cooling system.
Oil analysis offers information regarding the extent of this contamination and associated oil degradation status, so that changes in oil take place according to condition, not time. But what is the reason behind contamination of engine oil?
Most Common Contaminants
At the time of normal operation of an engine, a wide range of contaminants is introduced into the lubricating oil, as detailed below.
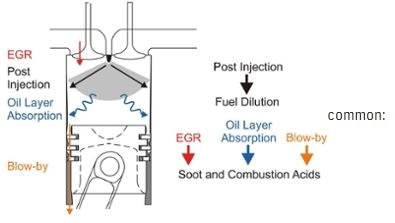
Combustion By-Products
Exhaust gases - or blowby gases - leak past the turbocharger seals, piston rings and valve guides into the crankcase and include particles of acids, carbon, partially burned fuels, water, lacquers, and varnish that contaminate the oil. Sulfur oxides (SOx) are common gases with sulfurous fuels (liquid fuel distillates, diesel, heavy fuel oil). Nitrous oxides (NOx) are found in higher concentrations in natural gas (LNG, CNG, propane) fueled engines. Hydrocarbon oxidation (HCOx) occurs in varying amounts.
Acids, Varnish, and Sludge
When the lubricating oil comes into contact with hot engine components, or when heated oil comes into contact with the entrapped air, oxidation and decomposition occur, thereby forming contaminants such as sludge, acids and varnish.
Fuel
Generally, these contaminants are associated with engine malfunction. However, fuel dilution can also be caused by stop-and-go operation and excessive engine idling and results in loss of viscosity, leading to severe wear and probable seizure if left unchecked. Fuel pump failure, faulty injectors, and clogged air filters are the most common reasons for fuel dilution; however, in certain cases, rupture of fuel lines could result in the contamination of oil.
Water
Water vapor is introduced into the oil as a combustion byproduct. Low load engines and backup generators prevent the oil from getting adequately hot to rapidly boil off the water. Acids formed as a result of combining water with blowby gases leads to degradation of the oil and corrosion of the engine surfaces. Ingress of water from broken cooling lines or the environment will result in rapid degradation of the oil, and could even lead to severe wear and engine failure.
Coolant
Glycol-based engine coolants are extensively used. Loss in integrity of cooling line fittings, head gasket seal rupture, and cooling system corrosion can all lead to mixing of the coolant with the oil. Particularly, glycol is corrosive to non-ferrous bearing surfaces. Excessive coolant results in the telltale sheen of "mayonnaise" oil emulsion, causing engine seizure.
Soot
Soot is formed due to the mixing of burning fuel with oil on the cylinder liner(s) or as a result of retarded injection timing. High amounts of soot cause abnormal valve and injector train wear and can overload the emissions control systems, leading to air quality fines.
Therefore, what are the tests to be performed for engine oil monitoring? Any test on engines must incorporate tests to search for common contaminants, and also tests to determine wear metals and oil viscosity.
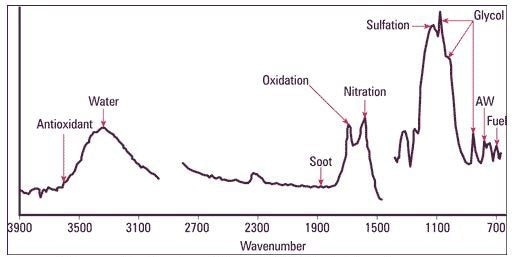
Typical used oil FTIR spectrum
Infrared spectroscopy is an excellent technique for detecting oil degradation products, water, and organic contaminants in a used oil sample. A single measurement gives abundant information in relation to the oil condition and contamination.
Oxidation and Nitration by Infrared Analysis
During severe oxidation, the lubricant corrodes the critical surfaces. The “oxidation number” governs the extent of oxidation. Conditions such as varnishing, sludge deposits, lacquering, sticky rings, and filter plugging occur in systems with oxidation problems.
Nitration is another significant parameter in detecting the quantity of NOx compounds in the oil. These compounds could react with water present in the oil and generate nitrous acids, thus degrading the oil and reducing the effectiveness of additives. This can form engine deposits, lacquer and varnish.
- Antiwear additive: Measured through IR, it is a measure of ZDDP additive that remains in the oil. ZDDP is an important friction reducing additive which leads to less protection against abnormal wear upon being exhausted.
- Soot: Measured through IR, it is a measure of the carbon present, in wt%. Even though most of the engine oils contain additives for dispersing soot, increasing amounts can overload its potential to do so, leading to wear and poor operation.
- Glycol content: It is a measure of the liquid glycol, in wt%. Glycol coolants break down under a high-temperature engine environment, leading to the generation of glycolic acids. These acids corrode non-ferrous bearing surfaces and result in the formation of metal salts. They also react with anti-oxidant and anti-wear additives in the oil (together with water) and form sludges that clog filters and destroy the lubricity properties of the oil, thus increasing abrasive wear. Glycol contamination in engines and transmissions is regarded as a more severe contaminant than water alone (up to 10 times more damaging). The temperature of the oil governs whether the glycol coolant disintegrates quickly or over time. Such an instability is a major challenge in ascertaining the actual glycol content in the oil at a given instant of time and is the predominant reason for the discrepancy between field and lab tests.
Viscosity
Viscosity is a fluid’s resistance to flow. Kinematic viscosity is the most significant physical property of engine lubricants. Generally, engine oils are multigrade formulations, designed to ensure normal operation of the engine over a broad range of startup operating temperatures. Usually, viscosity is reported at 100 °C. If a lubricant’s viscosity exceeds the ranges set by SAE for maximums and minimums, it is mandatory to change it. It is expected that oil viscosity increases over time and use with an increase in soot and solids. Loss in oil viscosity in an engine is regarded as more severe than the increase in viscosity and is generally caused by fuel contamination.

Total Base Number
Total base number (TBN) is the measure of the alkaline reserve in a lubricant, which is usually measured by titration, similar to the Karl Fischer technique. It can also be measured in other ways, for example, through infrared spectroscopy. Blowby gases, in combination with moisture from aggressive acids in the crankcase, deplete the base number. Additives added to engine oils are formulated to react with the acids formed and neutralize them. The base number is used as a guide to follow the depletion of additive of an oil in use. Oil changes are usually suggested if the TBN value reaches a preset level for a specific lubricant and application. A sudden decrease in TBN indicates abnormal operating conditions (for example, excessive blowby) that mandates investigation. A majority of the lubricant suppliers provide TBN condemnation limits in the bulletins. In general, if the TBN value is 30% of the original starting value, oil change is imminent. An oil with a high starting TBN value is recommended for engines that run on fuels with high sulfur content, and the alarm limit is 50% of the starting value, not 30%.
Water
One of the most common contaminants of oil is water, which can enter the engine oil as a combustion byproduct, or following a leak in the cooling system. Despite the fact that the engine oil temperature is adequately high to eliminate moisture during operation, elevated levels of water will induce damage wear. Excessive water that enters the engine as a result of ingression or leaks will lead to severe wear, resulting in engine seizure. Water contamination should never be more than 0.25% for a majority of the engines.
Total Acid Number
Total acid number (TAN) is a measure that indicates the relative acidity of a lubricant, which is usually measured by titration. The acid number is used as a guide to follow the oxidative degeneration of an oil in use. In engine applications, TAN is recommended for natural gas-fired engines. A sudden increase in TAN indicates abnormal operating conditions (for example, overheating ) that has to be investigated. A majority of the lubricant suppliers provide TAN condemnation limits in the bulletins. In general, a 2.0-point increase in the starting value is a cause for concern.
Ferrous Density
Ferrous density is a measure of the cumulative amount of ferrous magnetic debris in oil and is measured in ppm using a magnetometer. Ferrous debris that ranges in size from sub-micron to visible sizes leads to a variation in electrical current proportional to the amount of magnetic metal contained in oil. The total amount of ferrous debris is an important indicator of wear in any engine and should be incorporated on all screening test packages. The true value in ppm is trended and a 10% increase in the wear rate is indicative of an abnormal change that may necessitate intervention.

Fuel Dilution
Fuel dilution is a measure of the unburned liquid fuel that exists in the lubricant due to poor seals, leaking injectors, or poor combustion settings. All engines can tolerate some fuel; less than 2% is anticipated and more than 5% is a problem. Fuel dilution is measured by either SAW or GC sensor (fuel sniffer) and generally reported in wt%.
Particle Count
Particle count is a technique employed to count and categorize particulates in a fluid based on accepted size ranges, generally in accordance with ISO 4406 and SAE 4059. It is a very useful test to enhance reliability since decreasing particulates in the oil will increase the engine life. In general, it is an optional test for fielded engines as the soot content normally impedes an exact particle count. Particle count is usually used in engine development since technologies like LaserNet have the potential to effectively measure particle count in oils that contain up to 1.5% soot.
Ferrous Particle Count
Ferrous particle count is a method for quantifying the existing ferrous debris based on quantity and size, but not concentration. Intrinsic magnetometers (LNF or online) are widely used at present, and also the direct-read (DR) ferrography system. It is useful in detecting the size and quantity of the existing ferrous debris particles, which enhances the spectroscopy and ferrous density methods. The ratio of large and small particles in the sample is measured and the data can be used to calculate the severity indexes and the wear particle concentration, which may be alarmed.
Elemental Spectroscopy
Elemental spectroscopy is a method employed for the detection and quantification of metallic elements in a used oil caused by contamination, wear and additives. The oil sample is energized to drive each element to emit a quantifiable amount of energy, which denotes the concentration of the element in the oil. The results indicate the concentration of all dissolved metals, either in the form of particulates or from additive packages. This test is the base for all off-site and on-site oil analysis tools since it offers information on contamination, machine and wear condition in a comparatively accurate and rapid manner. Its major drawback is that its particle detection efficiency is poor for particles with a size of 5 μm or more, reiterating the salience of measuring the ferrous density first.
Wear Debris Analysis
Wear debris analysis (WDA) is defined as either an analytical or a patch technique that involves separating magnetic wear particles from the oil and depositing them on a glass slide called a ferrogram. Microscopic investigation of the patch or slide allows characterization of the wear mode and possible sources of wear in the machine. This method is called as analytical ferrography. Although it is an exceptional indicator of abnormal non-ferrous and ferrous wear, it is often performed only by a trained analyst.
The table below provides a summary of recommended test parameters.
| |
Viscosity |
TBN |
Water |
TAN |
Oxidation |
Nitration |
Elemental |
Soot |
Fuel Dilution |
Particle Count |
Ferrous Particle |
WDA |
| Engine Liquid |
R |
R |
R |
R |
R |
R |
R |
R |
R |
O |
O |
O |
| Engine NatGas |
R |
R |
R |
R |
R |
R |
R |
O |
N/A |
O |
O |
O |
R = Recommended; O = Optional

This information has been sourced, reviewed and adapted from materials provided by AMETEK Spectro Scientific.
For more information on this source, please visit AMETEK Spectro Scientific.